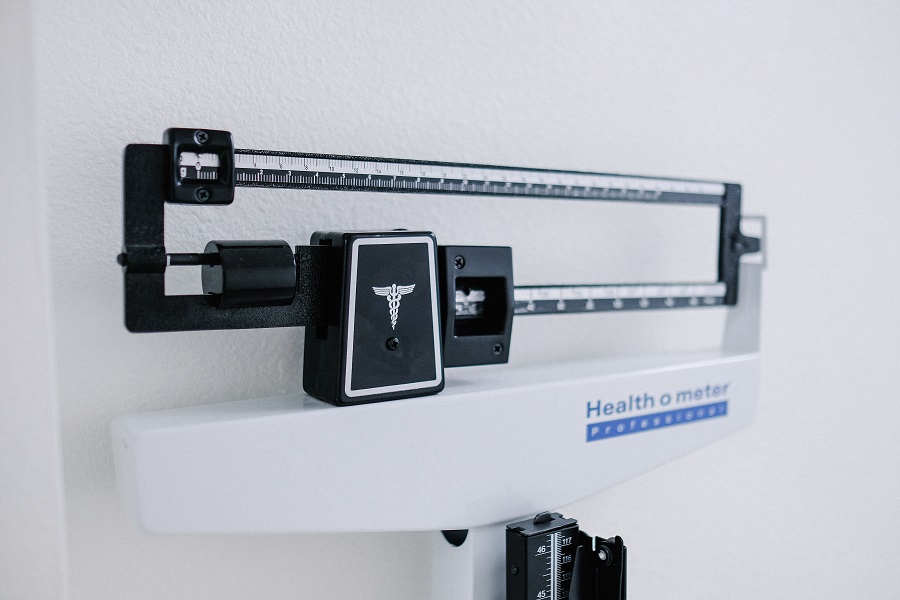
There’s a renewed focus among drugmakers on finding the most effective weight loss therapies. With a staggering 40% of adults in the U.S. classified as obese, the development of a successful pill for obesity presents a tremendous opportunity.
The global weight loss market is projected to skyrocket from US$224 billion in 2021 to more than US$405 billion by 2030, growing 6.84% annually. Racing to capture the bulk of the lucrative weight loss industry, these pharmaceutical companies are competing to roll out new obesity pills that are effective, convenient, and affordable, making them a better alternative to less appetizing injections.
These new oral weight management drugs are in various stages of clinical trials, holding the potential to generate hefty revenue when they hit the market.
Drug major Eli Lilly (LLY) develops and markets treatments in the fields of neuroscience, cardiometabolic, cancer, and immunology. While cancer, diabetes and immunology drugs are the key constituents of its product portfolio, the company also has a substantial and growing exposure to the obesity market.
“Lilly is well positioned to post industry-leading growth over the next five years, reinforcing its wide moat,” says a Morningstar sector director Damien Conover.
The high success rate of its injectable weight loss drug Mounjaro in phase 3 clinical trial prompted Conover to project peak annual sales estimate for the drug above US$18 billion.
More recently, Lilly is developing a weight-loss pill called orforglipron which has shown to be effective in its phase 2 trials. The oral version of GLP-1, a class of drug that replicates the actions of a naturally occurring hormone in the digestive system, orforglipron has also proven effective in the treatment of type 2 diabetes.
“Eli Lilly's innovative culture and strong financial commitment to developing the next generation of drugs set the company apart from its peers and fuel its long-term growth,” says Conover, who recently upped the stock’s fair value to US$368 from US$289 based on increased projections for diabetes and weight loss drugs Mounjaro and orforglipron.
Conover forecasts Mounjaro to become “Lilly's largest product over the next five years based on a leading efficacy profile in diabetes and weight loss.”
U.S. pharma giant Pfizer (PFE) sells healthcare products and chemicals. Prescription drugs and vaccines account for the majority of sales. The drugmaker sells its products globally, with international sales accounting for nearly half of its total sales.
“Pfizer's foundation remains solid, based on strong cash flows generated from a basket of diverse drugs,” says a Morningstar equity report.
Pfizer’s oral GLP-1, danuglipron, is in phase 2 clinical trials and the company is expected to finalize plans for a phase 3 program by the end of the year. A study found that high-dose versions of danuglipron twice a day was effective in reducing body weight of patients with Type 2 diabetes. The firm is concurrently working on developing a once-daily version of the drug. Pfizer CEO Albert Bourla recently said its obesity pill could eventually generate US$10 billion a year.
Pfizer, and other biopharma firms, could begin to launch their GLP-1-based obesity drugs as early as 2025, says Morningstar sector strategist, Karen Andersen.
The company's substantial scale underpins its robust competitive advantage when it comes to new drug development. “This unmatched heft, combined with a broad portfolio of patent-protected drugs, has helped Pfizer build a wide economic moat around its business,” says Andersen, who pegs the stock’s fair value at US$48, and forecasts close to 6% annual sales growth between 2020 and 2025 as new drugs offset generic competition.
Danish drugmaker, Novo Nordisk (NVO) is the leading provider of diabetes-care products in the world. Apart from capturing a third of the global branded diabetes treatment market, it manufactures and markets insulins, injectable diabetes treatments such as GLP-1 therapy, oral antidiabetic agents, and obesity treatments.
Novo Nordisk's highly popular Wegovy and Ozempic drugs, which created a surge of interest in the weight loss industry, are administered as weekly GLP-1 injections.
“Novo and Lilly will continue to lead the obesity market over the next 10 years, with incremental innovation in this rapidly expanding market that will support their wide moats,” says a Morningstar equity report.
Novo Nordisk's $1,350 per month Wegovy treatment, higher-dose oral and injectable semaglutide, and novel GLP-1/amylin cagrisema will support more than 35% share in 2032, says Andersen, who recently raised the stock’s fair value to US$130 from US$127 per ADR “after raising our assumed sales for the firm’s obesity portfolio and pipeline, as part of a broader, more bullish view on global obesity drug sales potential.”
Novo’s obesity sales could peak at US$23 billion by 2031, ahead of semaglutide’s 2032 patent expiration. “In obesity, semaglutide forms the basis of injectable obesity medicine Wegovy as well as a potential oral, high-dose semaglutide that recently generated positive data in a phase 3 trial,” says Andersen.
Wegovy's strong early uptake, and pipeline progress all point to Novo's ability to expand the drug-treated obesity market, she adds.




:quality(80)/cloudfront-us-east-1.images.arcpublishing.com/morningstar/22DGCGOHMJC7HHQIF2CERYV4JM.png)
:quality(80)/cloudfront-us-east-1.images.arcpublishing.com/morningstar/KEGKFRO24VF67PWV7V2ADNG37U.png)